What is NMN?
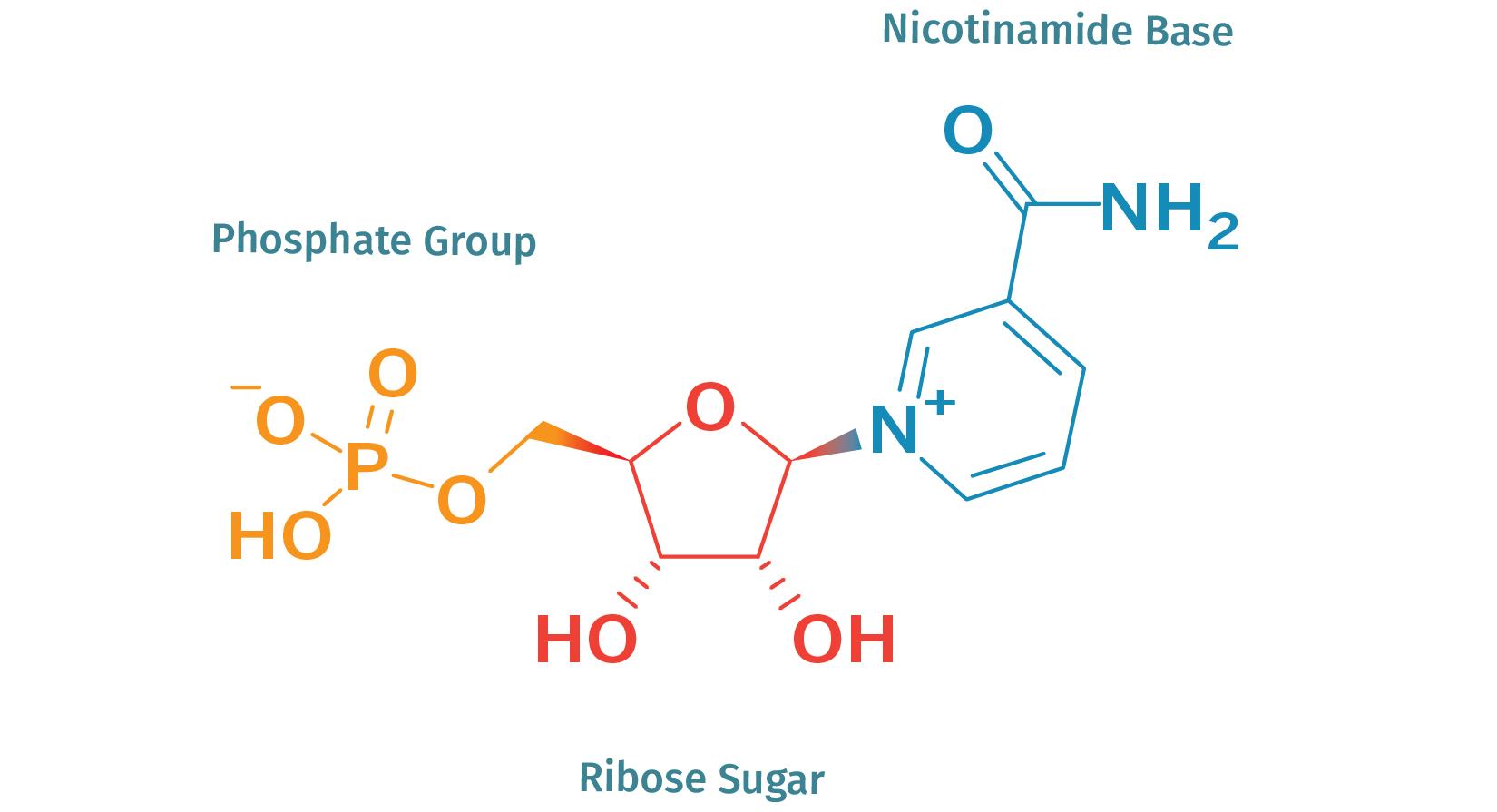
The acronym "NMN" stands for nicotinamide mononucleotide, a substance that is present in all living things. It's a ribonucleotide, which is a fundamental structural component of the nucleic acid RNA at the molecular level (Figure 1). Nicotinamide mononucleotide (NMN) is a type of nicotinamide adenine dinucleotide (NAD+) that serves as a precursor for this critical molecule in the body. It is important for raising NAD+ levels in cells.
What is Nicotinamide Adenine Dinucleotide (NAD+)?
The coenzyme NAD+ is a necessary component of life and cellular processes. Enzymes are catalyst molecules that enable chemical reactions to take place. Coenzymes are "assistants" that enzymes require in order to function.
What Does NAD+ Do?
The most prevalent molecule in the body after water, and without it, an organism would perish, is nicotinamide adenine dinucleotide (NAD+). Many proteins throughout the body use NAD+. It is essential for mitochondria, the cell's energy generators that produce chemical energy our bodies consume.
NAD+ Functions as a Coenzyme in Mitochondria
The enzyme nicotinamide adenine dinucleotide (NAD+) is a key regulator in metabolic processes including glycolysis, the TCA Cycle (AKA Krebs Cycle or Citric Acid cycle), and the electron transport chain, which takes place inside our mitochondria and provides cellular energy.
NAD+ serves as a ligand in which it binds to enzymes and transfers electrons between molecules. NAD+ acts through a cellular mechanism similar to recharging a battery by transferring electrons from one molecule to the next, much like as it does in its role as an enzyme-substrate.
When electrons are used to generate power, a battery is discharged. Those electrons can't go back to their starting point without assistance. NAD+ serves as a boost for those electrons in cells. As a result, NAD+ may lower or increase enzyme activity, gene expression, and cell signaling.
NAD+ Helps Control DNA Damage
DNA damage accumulates as organisms become older due to environmental influences such as radiation, pollution, and inaccurate DNA replication. The current aging theory holds that the accumulation of DNA damage is the primary cause of aging. The majority of cells have the ‘molecular machinery' needed to mend this damage. This equipment consumes NAD+ and energy molecules, as well as other biomolecules. As a result of this DNA damage, vital cellular resources might be depleted.
PARP (Poly (ADP-ribose) polymerase), one of the most important DNA repair proteins, is inhibited by NAD+ deficiency. Individuals over the age of 60 have lower levels of NAD+. PARP increases as a result of cellular damage from normal aging, decreasing NAD+ concentration. Any additional mitochondrial DNA damage adds to the problem.
PARP1 is ‘Middleman’ of DNA Repair
Schematic of How PARP1 Helps Repair Damaged DNA
How Does NAD+ Affect Sirtuins (the Longevity Genes) Activity?
Sirtuins, newly discovered proteins that act as “protectors of genes,” play an essential role in cell health. Sirtuins are a group of enzymes involved in cellular stress responses and damage repair.
There are also links between sirtuins and numerous other diseases, including diabetes, which is caused by insulin resistance. Sirtuins also control the aging process and disease states associated with it, such as neurodegenerative diseases and diabetes. NAD+ is required for sirtuin activation.
As David Sinclair, a Harvard geneticist and NAD researcher says we lose NAD+ as we age “and the resulting decline in sirtuin activity, is thought to be a primary reason our bodies develop diseases when we are old but not when we are young.” He believes that increasing NAD+ levels naturally while aging may slow or reverse certain aging processes.
Why Should We Care About NAD+
Since the discovery of NAD+ in 1906, scientists have been interested in the molecule due to its abundance within the body and important function in molecular pathways that keep our bodies functioning.
In animal studies, increasing NAD+ levels in the body has shown beneficial results in fields like metabolic and age-related illness, as well as anti-aging effects. Diabetes, cardiovascular disease, neurodegeneration, and general immune system loss are all examples of age-related illnesses.
Aging
Sirtuins, a family of enzymes that activate as we age, play an important role in maintaining genome integrity and aiding DNA repair. Like a car without gasoline will not operate, sirtuins' activation is dependent on NAD+. Results from animal studies suggest that raising NAD+ levels in the body activates sirtuins and extends yeast, worm, and mouse lifespans.
While animal research has shown promising results in anti-aging effects, scientists are still attempting to figure out how these findings might be applied to humans.
Metabolic Disorders
Excessive exercise depletes the body of NAD+, which is one of the key nutrients for healthy mitochondrial function and consistent energy production. The amount of NAD+ in the human body decreases as we age.
Niacin and nicotinamide, both forms of vitamin B3 (niacin), have also been shown in studies to help mice lose weight. Even in old mice, taking NAD+ boosters improved their exercise capacity and prevented diet-induced and age-related weight gain. Other research even reversed the diabetes impact in female mice, suggesting new approaches to tackle metabolic diseases like obesity.
Heart Function
NAD+ levels increase when you exercise and can be boosted through diet. High blood pressure can lead to an enlarged heart and blocked arteries, which can cause strokes.
NAD+ boosters have restored NAD+ levels in the hearts of mice and prevented injuries caused by a lack of blood flow. NAD+ boosters have also been shown to protect mice from abnormal heart growth.
Neurodegeneration
Increasing the NAD+ level in Alzheimer's mice decreased protein accumulation, which disrupted cell communication and improved cognitive function. When blood flow to the brain is limited, boosting NAD+ levels protects nerve cells from dying.
Many experiments in animal models have shown that consuming flavonoids may help to preserve brain health, ward off neurodegeneration, and boost memory.
Immune System
Adults become older, their immune systems weaken, and it becomes more difficult for them to recover from sickness. Seasonal viruses and even COVID-19 are becoming increasingly common.
Recent research has revealed that NAD+ levels are important in the regulation of inflammation and cell survival during inflammation and aging. The study suggested that NAD+ has therapeutic potential for immune dysfunctions.
How Does the Body Make Nicotinamide Adenine Dinucleotide (NAD+)?
Our bodies generate NAD+ from pre-existing components, or precursors. Consider them as the source of raw materials for NAD+. The following are five major precursors found in our bodies: L-tryptophan, Nam (nicotinamide), nicotinic acid (NA, or niacin), nicotinamide riboside (NR), and nicotinamide mononucleotide (NMN). Of these, NMN represents one of the final steps of NAD+ synthesis.
The body converts some precursors into other chemicals. Vitamin B3 is a necessary nutrient that may be found in several foods. Various pathways within the body allow our cells to create NAD+ from niacinamide, NA, and NR. A biochemical pathway is comparable to a manufacturing line at work in the factory. In the case of NAD+, many production lines result in the same product.
The de novo path is the first of these options. The term de novo means "from scratch" in Latin. The de novo pathway begins with tryptophan, which is the earliest of the NAD+ intermediates, and proceeds upward from there.
The salvage pathway is the second method. The salvaging mechanism, like recycling, converts NAD+ generated by the degradation of molecules into NAD+. All proteins in the body must be degraded on a regular basis to prevent them from accumulating too much.
Enzymes operate in the same way. They consume some of the results of a protein's breakdown and return it to the production line.
NAD+ Biosynthesis from NMN
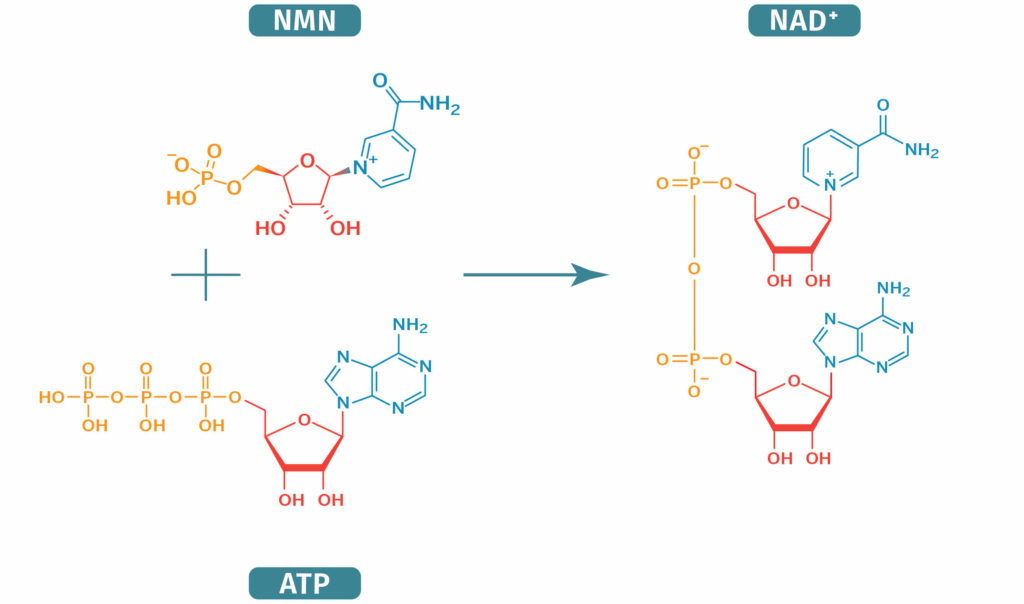
How is NMN Synthesized In the Body?
B vitamins are required to produce NMN. In the body, nicotinamide phosphoribosyltransferase (NAMPT) is the enzyme that converts nicotinamide (vitamin B3) to NMN. NAMPT links nicotinamide (a vitamin B3) to PRPP (5'phosphoribosyl-1-pyrophosphate), which is a sugar-phosphate. NMN can also be made from ‘nicotinamide riboside’ (NR) through the addition of a phosphate group.
‘NAMPT' is a rate-limiting enzyme in the synthesis of NAD+. This means reduced levels of NAMPT result in lower NMN production and lowered NAD+ levels. precursor molecules such as NMN may also boost NAD+ synthesis by adding them to the mix.
Methods to Increase NAD+ levels
Calorie restriction, also known as calorie reduction, has been linked to improved NAD+ levels and sirtuin activity. Calorie restriction in mice has been found to slow aging. Although NAD+ is found in certain foods, the amounts are insufficient to change intracellular NAD+ levels. It has been shown that taking particular supplements, such as NMN, can boost NAD+ levels.
NAD Supplement as NMN
Intracellular levels of NAD+ decline as normal cellular activities consume up NAD+ reserves with age. Supplementation with NAD+ precursors is believed to aid in the recovery of healthy levels of NAD+. According to the study, precursor molecules such as NMN and nicotinamide riboside (NR) are considered supplements for increasing NAD+ levels.
David Sinclair, an NAD+ researcher from Harvard, says, “Feeding or administering NAD+ directly to organisms is not a practical option. The NAD+ molecule cannot readily cross cell membranes to enter cells, and therefore would be unavailable to positively affect metabolism. Instead, precursor molecules to NAD+ must be used to increase bioavailable levels of NAD+.”
NAD+ cannot be used as a direct supplement because it is not readily absorbed. NAD+ precursors are more easily absorbed and more effective supplements than NAD+.
How are NMN Supplements Absorbed and Distributed Throughout the Body?
NMN is absorbed via a cell membrane-bound transporter, which appears to be embedded in the cell surface. Because the NMN molecule is smaller than NAD+, it may be more easily absorbed into cells. The waterless area of the cellular membrane prevents ions, polar molecules, and big molecules from entering without the need for transporters because it is impermeable.
It was previously believed that NMN must be changed before it enters cells, but new research suggests that it may enter cells directly via an NMN-specific transporter on the cellular membrane.
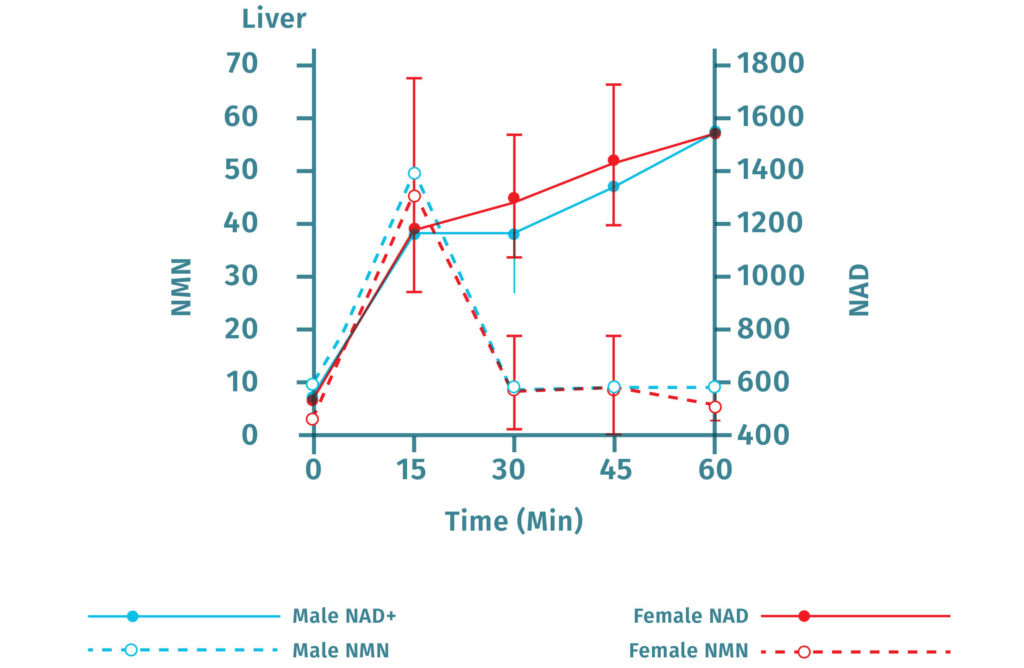
Furthermore, NMN injections result in increased NAD+ levels in a number of tissues, including the pancreas, fat tissue, heart, skeletal muscle, kidneys, testes, eyes, and blood vessels. Within 15 minutes of oral administration of NMN in mice, NAD+ levels increase in the liver.
NMN is Quickly Converted to NAD+
NMN Side Effects and Safety
Cis-1,3-dienoylshbetacyanin (DMN) is considered safe in animals and the findings are encouraging enough that human clinical trials have begun. Even at high dosages, this chemical is generally thought to be non-toxic and not hazardous. In mice, long-term (one-year) oral administration does not result in any toxic effects.
The first clinical trial in people was completed, and the data back it up: it is not harmful in single doses.
However, following NMN ingestion, Japanese males had increased levels of bilirubin in their blood, which remained within the typical range. Future studies should concentrate on long-term safety and effectiveness. NMN does not appear to have any other adverse effects.
The History of NMN and NAD+
Nicotinamide adenine dinucleotide, often known as NAD, is one of the most essential and versatile molecules in the human body. Because it is necessary for cells to consume energy, almost no biological activity takes place without it. As a result of this, NAD has garnered a significant amount of attention from researchers throughout the world.
1906
In 1906, Arthur Harden and William John Young discovered a "factor" in liquid drawn from brewer's yeast that increased the conversion of sugar to alcohol during fermentation. NAD was found to be the cause of this "coferment."
1929
Harden, along with Hans von Euler-Chelpin, continued to unravel the secrets of fermentation. They were given the Nobel Prize in 1929 for their research into these processes, which included determining the chemical form and properties of NAD.
During the 1930s, under the direction of Otto Warburg, another Nobel laureate, who identified NAD's key function in many biochemical processes, the account of NAD grew. The transfer of electrons from one atom to another is the source of all biological activities' required energy.
1937
In 1937, Conrad Elvehjem and colleagues at the University of Wisconsin, Madison, discovered that NAD+ treatment cured pellagra in dogs. Pellagra is a disease caused by a niacin deficit that manifests itself as diarrhea, dementia, and ulcers in the mouth. It results from niacin insufficiency and is now generally treated with nicotinamide (one of the NMN precursors).
The work by Arthur Kornberg on NAD+ in the 40s and 50s helped him to discover the principles behind DNA replication and RNA transcription, two vital life processes.
1958
In 1958, Jack Preiss and Philip Handler discovered the three biochemical phases by which nicotinic acid is converted to NAD. The Preiss-Handler Pathway is a sequence of events known today as the Preiss-Handler Pathway.
In the same year, Chambon, Weill, and Mandel discovered that nicotinamide mononucleotide (NMN) provided the energy needed to activate an important nuclear enzyme. This finding paved the way for a slew of amazing findings of a class of proteins known as PARPs, including their role in DNA damage repair, cell death regulation, and lifespan variation.
1976
Rechsteiner and his colleagues discovered convincing evidence in 1976 that NAD+ had a "possibly other essential function" in mammalian cells, beyond its classic biochemical function as an energy transfer molecule.
Because of this, Leonard Guarente and his coworkers were able to discover that sirtuins use NAD to extend their life span by keeping certain genes "shut."
Since then, interest has developed in NAD and its metabolites, NMN and NR, for their antiaging properties.
The Future of Nicotinamide Mononucleotide
Researchers are working to figure out how NMN works in the human body with the potential therapeutic effects that it has shown in animal trials. The molecule was found safe and well-tolerated at the given dosage during a recent clinical trial in Japan. More studies and human trials are planned. It's an interesting and adaptable molecule with which we still have much to learn from.







